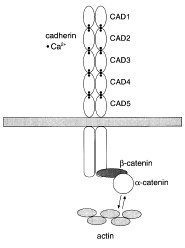
Schematic
Representation
Classic
cadherin molecules at the cell surface
|
Cadherins
are differentially expressed during development
and in adult organs. Since many cell types
express multiple cadherin subclasses simultaneously
(the combination differs with cell type),
it can be inferred that the adhesion properities
of individual cells are thus governed by
varying the combinations of cadherins. There
is also recent evidence to suggest that
altered expression of cadherins may be involved
in invasion and metastasis of tumour cells.
On
the cell surface, cadherins tend to be concentrated
at cell-cell junctions. Here they are structurally
associated with cortical actin bundles.
The cytoplasmic domain has been shown to
be associated with cytoplasmic proteins
termed catenins. Deletion of the cytoplasmic
domain destroys these interactions and also
eliminates cadherin function. Recently src,
yes, and lyn gene products of the src proto-oncogene
family have been found expressed at cell-cell
junctions. These kinases may be responsible
for cadherin phosphorylation and this raises
the possibly that cadherin mediated cell
junctions might be used for intercellular
signalling
|